
Stability of a Series of BODIPYs in Acidic Conditions: An Experimental and Computational Study into the Role of the Substituents at Boron | ACS Omega

Sahovsky - Enciclopaedia Del Medio Juego Problemasinformator PDF | PDF | Abstract Strategy Games | Traditional Board Games

Mechanism and the Origins of Periselectivity in Cycloaddition Reactions of Benzyne with Dienes | The Journal of Organic Chemistry
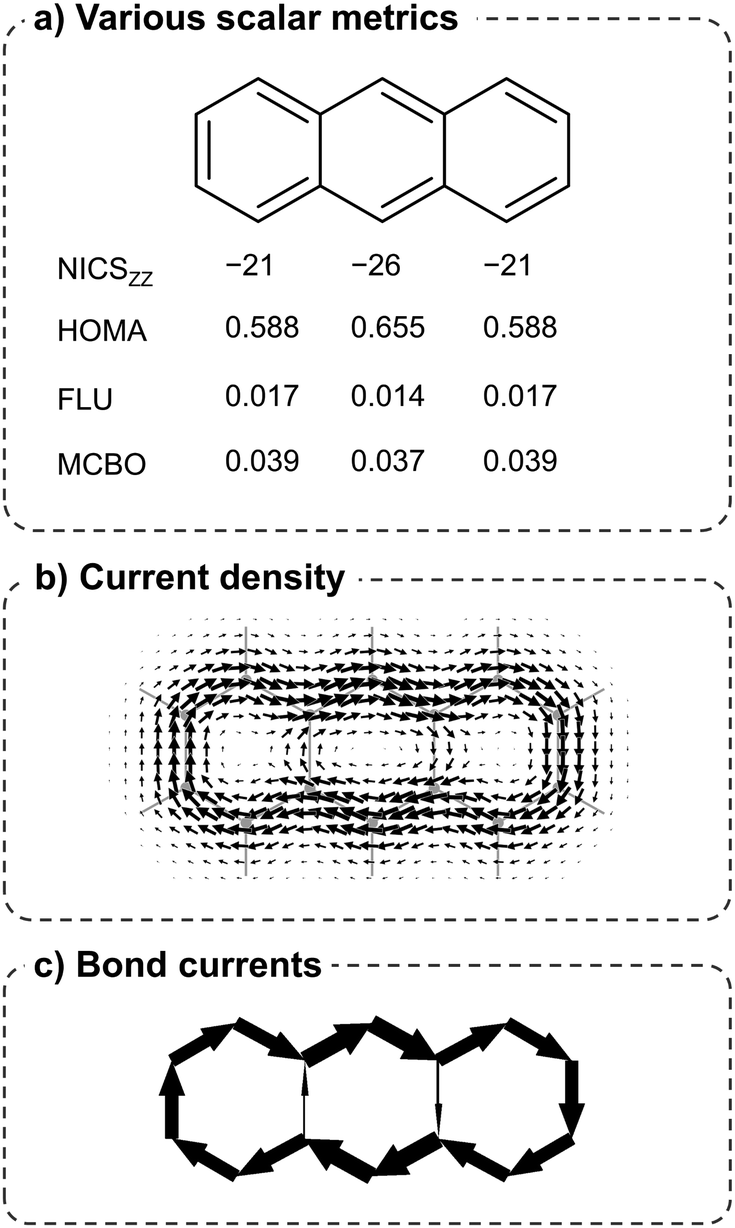
Simple and efficient visualization of aromaticity: bond currents calculated from NICS values - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D1CP05757J

Perimeter Coordinated Diastereomeric Rh(I) Complex of Helically Twisted Weakly Aromatic Hybrid Singly N-Confused β–β Fused Ferrocenoporphyrinoids | The Journal of Organic Chemistry












